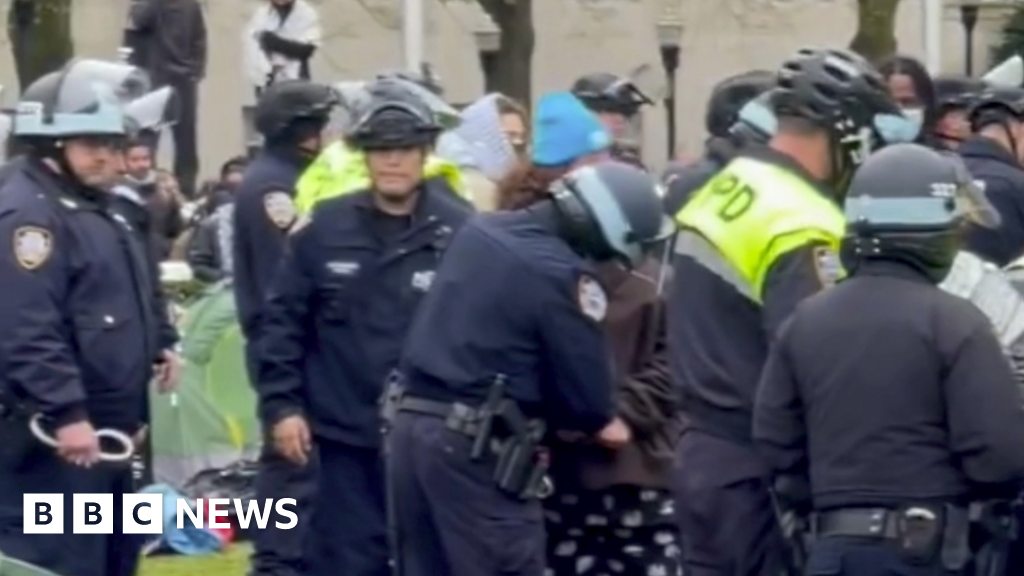U.K.
Rishi Sunak vows to end Britain’s ‘sick note culture’
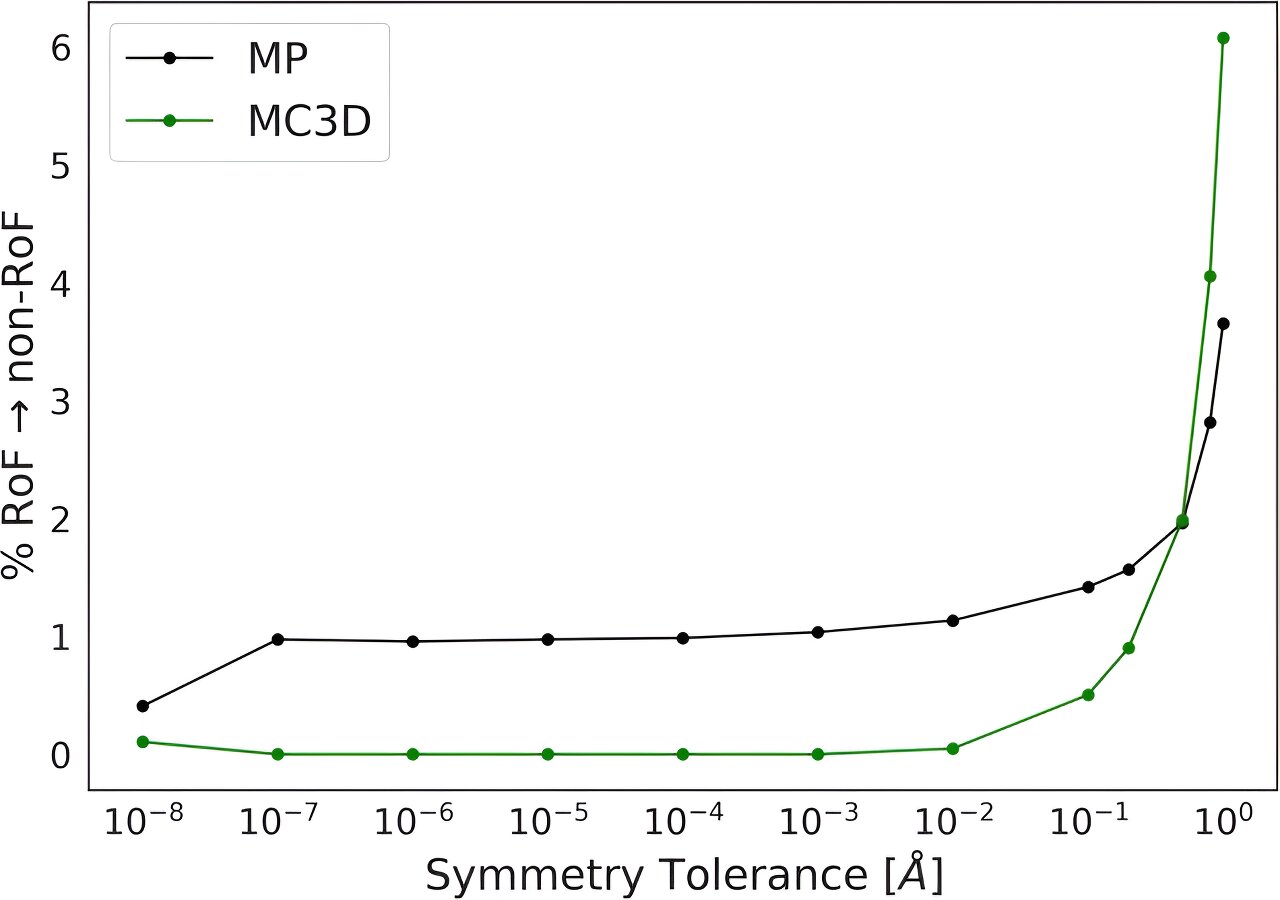
Materials follow the ‘Rule of Four,’ but scientists don’t know why yet
Science
This article has been reviewed according to Science X’s editorial process and policies. Editors have highlighted the following attributes while ensuring the content’s …
Covid warning with cases rising once again – UKHSA scientists send urgent plea to public
Health
Brits who are infected with Covid are being asked to stay at home or use a face mask if they have to …
Infected blood scandal: Children were used as ‘guinea pigs’ in clinical trials
U.K.
By Chloe Hayward and Hugh Pym Health producer and health editor, BBC News 2 hours ago Image source, Allan Archive Image caption, …
US blocks Palestinian push for full UN membership at Security Council | United Nations News
World
The United States has vetoed a widely supported resolution at the UN Security Council (UNSC) that would have paved the way for …
Badenoch attacks diversity red tape for stifling Britain’s growth
Business
Kemi Badenoch has attacked plans by the City regulator to set diversity targets for finance companies, arguing the proposals are counterproductive and …
The British scientists plotting to control the world’s weather
Technology
The Reading scientists, led by Prof Giles Harrison, have meanwhile been developing a different technique, in which an electric charge is applied …
Prince Harry ‘trolling’ King Charles with USA residency: ‘Bitter over Frogmore!’
Entertainment
Prince Harry has been accused of “trolling” King Charles, after choosing a specific date on his business documents to mark his US …
Hammers gave everything against a team that appear to be invincible as Xabi Alonso’s side reach Europa League semi-finals
Sports
Michail Antonio’s early goal inspired hope but West Ham couldn’t level the tie Jeremie Frimpong struck late to seal a 3-1 aggregate …